https://butterlab.imb-mainz.de/flydev/
Published in Genome Research.
https://genome.cshlp.org/content/27/7/1273.short
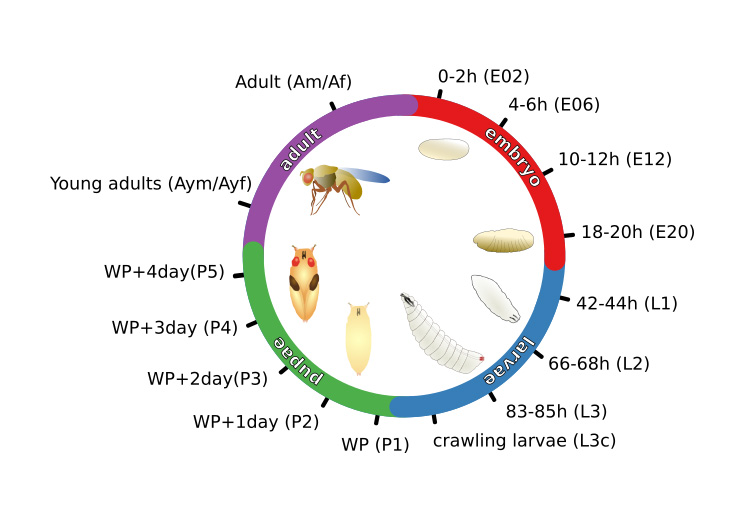
Drosophila melanogaster is a widely used genetic model organism in developmental biology. While this model organism has been intensively studied at RNA level, a comprehensive proteomic study covering the complete life cycle is still missing. Here, we apply label-free quantitative proteomics to explore proteome remodeling across Drosophila’s life cycle, resulting in 7,952 proteins, and provide a high temporal-resolved embryogenesis proteome of 5,458 proteins. Our proteome data enabled us to monitor isoform-specific expression of 34 genes during development, to identify the pseudogene Cyp9fpsi as a protein-coding gene and to obtain evidence of 268 small proteins. Moreover, the comparison with available transcriptomic data uncovered examples of poor correlation between mRNA and protein, underscoring the importance of proteomics to study developmental progression. Data integration of our embryogenesis proteome with tissue-specific data revealed spatial and temporal information for further functional studies of yet uncharacterized proteins. Overall, our high resolution proteomes provide a powerful resource and can be explored in detail in our interactive web interface.
